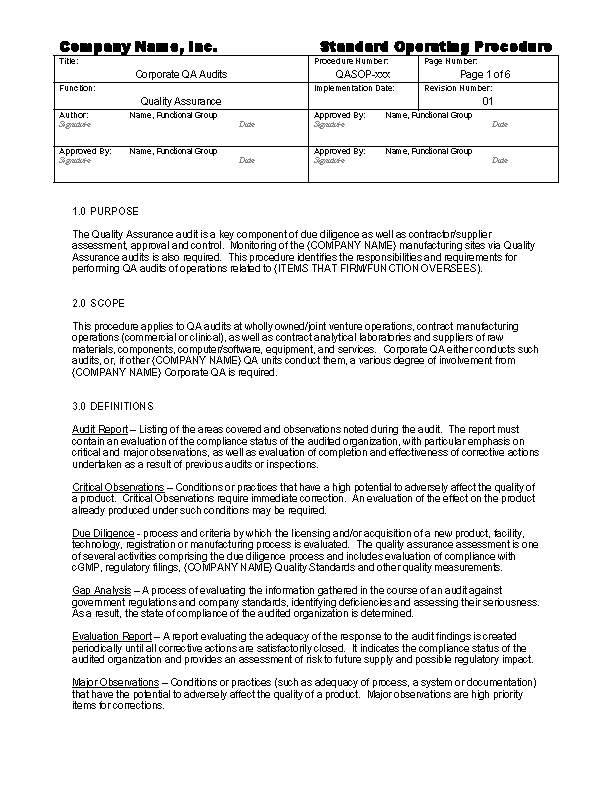
Batch Record Review
$39.95
Batch records are commonly also known as: production records, manufacturing records. Whatever your firm calls them, you need a procedure on how review such records and record that review. This procedure describes how to effectively review batch records and document the review on the appropriate forms. During the review of batch records, it’s helpful to have a checklist to guide you along the way.
Certificate of Analysis Package
$9.95
Certificates of Analysis are required documents for most batches to be QA Released by an organization. This product contains 2 templates for a Certificate of Analysis. The first document is just a blank template/shell for a properly and cleanly formatted CofA. The second document is a filled in example template with example tests and result specifications for a typical biopharmaceutical product.
Post Marketing Surveillance / Recalls
$39.95
This document describes the procedure for performing post marketing surveillance and market actions (i.e., recalls) affecting products cleared for commercial distribution by the firm. It creates the requirements and sets the responsibilities for appropriate functional groups during a product recall.
QA Hold System
$39.95
This document describes the steps for initiating, processing, and closing Quality Assurance (QA) Hold Reports associated with suspect non-conforming product or material. It allows for the quarantine of materials so that they can be investigated and given final disposition (approve or reject). This package includes the standard operating procedure (SOP) as well as the associated QA Hold Report form.
Raw Material Receipt and Disposition
$39.95
This document describes the process for the receipt, disposition, and release of raw materials used in or supporting GMP production at a manufacturing facility. It covers the controls necessary for proper material flow and labeling as well. This document can apply for pharmaceutical or medical device manufacturing firms.
Reagent and Powder Filling / Labeling Procedure
$19.95
This package contains two separate SOPs that work in conjunction with each other. The first document is a general reagent and powder filling procedure. It is appropriate for most manufacturers of chemicals, medical devices, or pharmaceuticals that have reagent or powder filling operations. The second document is a hardware-specific SOP for the operation of an automated powder filling system (the Hierath & Andrews ISO-G 4104 Direct-Weight Filler). Note that these documents need to be customized and tailored to your specific operations, but do provide an excellent template/example for how these documents should be drafted.